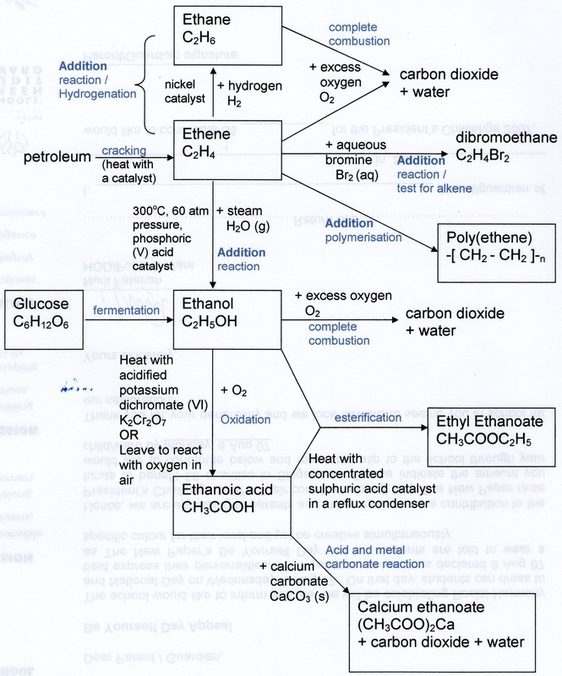
Summary of important organic chemistry reactions
Supplementary notes to diagram
Do note the chemical equation for fermentation of glucose is
C6H12O6 → 2C2H5OH + 2CO2. Carbon dioxide as a product in this reaction is not shown in the diagram. Aqueous solution of glucose, yeast to provide enzyme catalyst and suitable temperature of 37˚C are needed conditions.
Do note that the chemical equation for oxidation of ethanol to ethanoic acid when ethanol is left in air is
C2H5OH + O2 → CH3COOH + H2O. The product water is not shown in the diagram but is actually produced.
Do note that the chemical equation for esterification in the diagram is
CH3COOH + C2H5OH → CH3COOC2H5 + H2O. The product water is not shown in the diagram but is actually produced.
In stating the oxidising agent used in oxidation of alcohol to carboxylic acid, it is better to state as "acidified potassium dichromate (VI) containing dilute sulphuric acid". It is better to state the name of the acid that is used here (which is sulphuric acid).
Do note the chemical equation for fermentation of glucose is
C6H12O6 → 2C2H5OH + 2CO2. Carbon dioxide as a product in this reaction is not shown in the diagram. Aqueous solution of glucose, yeast to provide enzyme catalyst and suitable temperature of 37˚C are needed conditions.
Do note that the chemical equation for oxidation of ethanol to ethanoic acid when ethanol is left in air is
C2H5OH + O2 → CH3COOH + H2O. The product water is not shown in the diagram but is actually produced.
Do note that the chemical equation for esterification in the diagram is
CH3COOH + C2H5OH → CH3COOC2H5 + H2O. The product water is not shown in the diagram but is actually produced.
In stating the oxidising agent used in oxidation of alcohol to carboxylic acid, it is better to state as "acidified potassium dichromate (VI) containing dilute sulphuric acid". It is better to state the name of the acid that is used here (which is sulphuric acid).