*Please click video title headings to play video if video does not load properly.
Pressure
Pressure is the force acting per unit area whereby the force is applied in a perpendicular direction to the surface of an object.
Pressure = Force / Area , P = F / A
S.I. unit for pressure is pascal (Pa) or N m-².
Pressure increases when force applied increases.
Pressure increases when area where force is acting decreases.
Examples where concept of pressure are used:-
1. Knife is made sharp at its edge to have small area and thus a large pressure is applied when the person cuts something using the knife.
2. Lorries that carry heavy loads have many wheels. This increases surface area in contact with road and reduces pressure on the road.
Pressure = Force / Area , P = F / A
S.I. unit for pressure is pascal (Pa) or N m-².
Pressure increases when force applied increases.
Pressure increases when area where force is acting decreases.
Examples where concept of pressure are used:-
1. Knife is made sharp at its edge to have small area and thus a large pressure is applied when the person cuts something using the knife.
2. Lorries that carry heavy loads have many wheels. This increases surface area in contact with road and reduces pressure on the road.
Pressure = Force / Area (Bed of nails experiment)
See following video which demonstrates that a ballon can be easily pierced by one single nail as the force is acting through a small area (one nail) onto the ballon, creating a large pressure on the balloon causing it to burst. However, if the same amount of force is applied over a bed of many nails onto the balloon, the pressure on the balloon is now smaller and it is harder to cause the balloon to burst.
Transmission of pressure in incompressible liquids
Pressure is transmitted with equal magnitude in all directions through an incompressible liquid.
This concept of transmission of pressure through incompressible liquid is used in a hydraulic press (used to lift heavy objects such as car) or hydraulic brake system (used in car brakes to stop a car).
See following two videos which explore the concepts of hydraulic press and hydraulic brake systems.
This concept of transmission of pressure through incompressible liquid is used in a hydraulic press (used to lift heavy objects such as car) or hydraulic brake system (used in car brakes to stop a car).
See following two videos which explore the concepts of hydraulic press and hydraulic brake systems.
Hydraulic press system
More details on hydraulic press system
Note that left side piston has a very small area while the right side piston has a much larger area. But the pressure transmitted from left piston is equal to pressure received on right piston since the liquid inside is incompressible and pressure is transmitted equally in all directions through the liquid.
Thus, P1 = P2 , P1 is pressure transmitted by left piston, P2 is pressure received by right piston.
F1 / A1 = F2 / A2 , using the formula P = F / A.
Qn 1a: Let's say the hydraulic press has left piston cross-sectional area of 50 cm³ and right piston cross-sectional area of 500 cm³. It is used to lift an object of weight 1000 N. How much force is needed to push down on the left piston to lift the object placed on right piston?
P1 = P2
F1 / A1 = F2 / A2
F1 / 50 = 1000 / 500
F1 = 100 N
From the calculations, we can see that it requires a much smaller force of 100 N applied at left piston to lift a heavy object of 1000 N placed at right piston.
Qn 1b: Let's say the right piston is moved upwards by a height of 15 cm. What is the distance the left piston has to move downwards to produce this upward movement of right piston?
Since volume of liquid displaced by left piston (V1) is equal to volume of liquid gained (V2) to move right piston upwards as the liquid is incompressible,
V1 = V2
A1h1 = A2h2 , using the formula of volume of cylinder V = Base area (A) X height (h)
50 X h1 = 500 X 15
h1 = 150 cm
Thus, the left piston has to move down a larger distance of 150 cm just to lift up the heavy object on the right piston by only 15 cm.
In the following video below, it uses another method to calculate the distance moved downwards by the left piston using the concept of conservation of energy.
Work done by left piston = Work done by right piston
F1 X h1 = F2 X h2
100 X h1 = 1000 X 15
h1 = 150 cm
Notice both methods whether using V1 = V2 or Work done by left piston = Work done by right piston give the same answer for h1. This is the beauty of scientific theories if they are already proven to be correct theories, they should work in the circumstances they are supposed to work in.
Thus, P1 = P2 , P1 is pressure transmitted by left piston, P2 is pressure received by right piston.
F1 / A1 = F2 / A2 , using the formula P = F / A.
Qn 1a: Let's say the hydraulic press has left piston cross-sectional area of 50 cm³ and right piston cross-sectional area of 500 cm³. It is used to lift an object of weight 1000 N. How much force is needed to push down on the left piston to lift the object placed on right piston?
P1 = P2
F1 / A1 = F2 / A2
F1 / 50 = 1000 / 500
F1 = 100 N
From the calculations, we can see that it requires a much smaller force of 100 N applied at left piston to lift a heavy object of 1000 N placed at right piston.
Qn 1b: Let's say the right piston is moved upwards by a height of 15 cm. What is the distance the left piston has to move downwards to produce this upward movement of right piston?
Since volume of liquid displaced by left piston (V1) is equal to volume of liquid gained (V2) to move right piston upwards as the liquid is incompressible,
V1 = V2
A1h1 = A2h2 , using the formula of volume of cylinder V = Base area (A) X height (h)
50 X h1 = 500 X 15
h1 = 150 cm
Thus, the left piston has to move down a larger distance of 150 cm just to lift up the heavy object on the right piston by only 15 cm.
In the following video below, it uses another method to calculate the distance moved downwards by the left piston using the concept of conservation of energy.
Work done by left piston = Work done by right piston
F1 X h1 = F2 X h2
100 X h1 = 1000 X 15
h1 = 150 cm
Notice both methods whether using V1 = V2 or Work done by left piston = Work done by right piston give the same answer for h1. This is the beauty of scientific theories if they are already proven to be correct theories, they should work in the circumstances they are supposed to work in.
Hydraulic brake system
Pressure at different heights in a column of liquid
Pressure due to a column of liquid = height of column of liquid X density of liquid X gravitational field strength
P = hρg
See following video for how this formula is derived and also a simple calculation of pressure using this formula. It is not necessary to know how to derive this formula though I will suggest to be aware of it so that the understanding of this formula is better internalised.
P = hρg
See following video for how this formula is derived and also a simple calculation of pressure using this formula. It is not necessary to know how to derive this formula though I will suggest to be aware of it so that the understanding of this formula is better internalised.
Pressure measurement using mercury barometer
At sea level, the atmospheric pressure is approximately 1.03 X 10^5 Pa.
Atmospheric pressure can be measured using a mercury barometer. The height of mercury in a mercury barometer is 760 mm. Thus, atmospheric pressure is 760 mmHg or 76 cmHg.
P = hρg
1.03 X 10^5 = h(13600)(10) , P is the atmospheric pressure, ρ of mercury is 13600 kg m-³ , g is 10 ms-².
h = 0.757 m
= 76 cm
= 760 mm
Doing the simple calculation above, we can tell that the height of mercury in a mercury barometer is always approximately 76 cm or 760 mm at one atmospheric pressure (1.03 X 10^5 Pa).
It does not matter whether the mercury barometer is tilted or vertical, the vertical height of the column of mercury is always 76 cm (unless it is tilted so far downwards below vertical height of 76 cm that the whole barometer is now completely filled with mercury).
See following video for the same calculation done for a mercury barometer.
Atmospheric pressure can be measured using a mercury barometer. The height of mercury in a mercury barometer is 760 mm. Thus, atmospheric pressure is 760 mmHg or 76 cmHg.
P = hρg
1.03 X 10^5 = h(13600)(10) , P is the atmospheric pressure, ρ of mercury is 13600 kg m-³ , g is 10 ms-².
h = 0.757 m
= 76 cm
= 760 mm
Doing the simple calculation above, we can tell that the height of mercury in a mercury barometer is always approximately 76 cm or 760 mm at one atmospheric pressure (1.03 X 10^5 Pa).
It does not matter whether the mercury barometer is tilted or vertical, the vertical height of the column of mercury is always 76 cm (unless it is tilted so far downwards below vertical height of 76 cm that the whole barometer is now completely filled with mercury).
See following video for the same calculation done for a mercury barometer.
Introduction to atmospheric pressure and manometers
See following video on discussion of why atmospheric pressure increases as one goes to lower altitudes and decreases as one goes to higher altitudes. Just imagine how many air molecules are pressing down on oneself. As one heads to lower altitudes, he experiences more air molecules pressing down on him while as he goes to higher altitudes, there are lesser air molecules there which press down on him, so he experiences lower atmospheric pressure.
He will also find it harder to breathe at higher altitudes since the low atmospheric pressure makes the difference in air pressure between the atmosphere and his lungs less significant and it is more difficult for air to enter his lungs due to the lower difference in air pressure. Also, lesser air molecules at higher altitudes mean lesser oxygen that can enter his lungs and be carried in his bloodstream to various body tissues, so he will have tendency of fainting spells and cannot engage in strenuous activities due to lack of oxygen reaching his various body tissues.
We can use manometer to measure atmospheric pressure as well as pressure of gases. The mercury barometer is one example of a closed up manometer which has a vacuum inside used to measure atmospheric pressure.
He will also find it harder to breathe at higher altitudes since the low atmospheric pressure makes the difference in air pressure between the atmosphere and his lungs less significant and it is more difficult for air to enter his lungs due to the lower difference in air pressure. Also, lesser air molecules at higher altitudes mean lesser oxygen that can enter his lungs and be carried in his bloodstream to various body tissues, so he will have tendency of fainting spells and cannot engage in strenuous activities due to lack of oxygen reaching his various body tissues.
We can use manometer to measure atmospheric pressure as well as pressure of gases. The mercury barometer is one example of a closed up manometer which has a vacuum inside used to measure atmospheric pressure.
Calculations involving manometers
I provide below a few simple examples of calculations involving closed and opened manometers for your reference. Do note that there are even more complex problems than these, but the concepts used in the calculations are always similar.
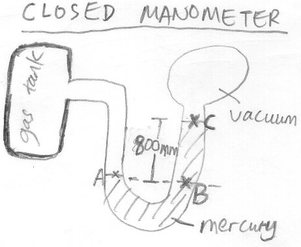
Qn 1: Find the gas pressure in the gas tank. (Density of mercury = 13600 kg m-³)
Note that pressures at same level are always the same.
Thus, PA = PB
PA = PC + Pressure difference between B and C
At point B, we need to account for the pressure at point C plus the entire column of mercury pressing down on point B which is the pressure difference between B and C.
Therefore, PA = 0 + hρg , PC is 0 Pa since there is no pressure at point C due to the vacuum.
= (0.8)(13600)(10) , hρg is being used to find the pressure at point B due to mercury column
= 1.08 X 10^5 Pa (all units in the calculation are in SI units)
Gas pressure is thus 1.08 X 10^5 Pa.
Note that pressures at same level are always the same.
Thus, PA = PB
PA = PC + Pressure difference between B and C
At point B, we need to account for the pressure at point C plus the entire column of mercury pressing down on point B which is the pressure difference between B and C.
Therefore, PA = 0 + hρg , PC is 0 Pa since there is no pressure at point C due to the vacuum.
= (0.8)(13600)(10) , hρg is being used to find the pressure at point B due to mercury column
= 1.08 X 10^5 Pa (all units in the calculation are in SI units)
Gas pressure is thus 1.08 X 10^5 Pa.
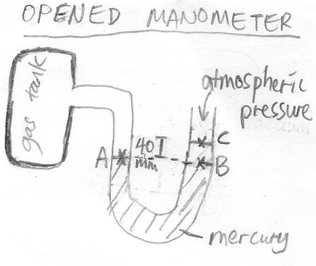
Qn 2: Find the gas pressure in the gas tank. Note that in this new question, the same gas is still used just that the manometer is an opened type manometer, so it is exposed to atmospheric pressure.
Note that pressures at same level are always the same.
Thus, PA = PB
PA = PC + Pressure difference between B and C
At point B, we need to account for the pressure at point C plus the entire column of mercury pressing down on point B which is the pressure difference between B and C.
Therefore, PA = 1.03 X 10^5 + hρg , PC is 1.03 X 10^5 Pa since there is atmospheric pressure at point C due to atmosphere.
= 1.03 X 10^5 + (0.04)(13600)(10) , hρg is being used to find the pressure at point B due to mercury column
= 1.08 X 10^5 Pa (all units in the calculation are in SI units)
Gas pressure is thus 1.08 X 10^5 Pa.
From the above two questions, we can see that the same gas should give same gas pressure measured be it using the closed or opened type manometers provided one does the calculations for both cases correctly using knowledge of pressure. Again, this is the beauty of science theories that are proven to be correct almost all the time unless refuted by newer findings and discoveries.
Note that pressures at same level are always the same.
Thus, PA = PB
PA = PC + Pressure difference between B and C
At point B, we need to account for the pressure at point C plus the entire column of mercury pressing down on point B which is the pressure difference between B and C.
Therefore, PA = 1.03 X 10^5 + hρg , PC is 1.03 X 10^5 Pa since there is atmospheric pressure at point C due to atmosphere.
= 1.03 X 10^5 + (0.04)(13600)(10) , hρg is being used to find the pressure at point B due to mercury column
= 1.08 X 10^5 Pa (all units in the calculation are in SI units)
Gas pressure is thus 1.08 X 10^5 Pa.
From the above two questions, we can see that the same gas should give same gas pressure measured be it using the closed or opened type manometers provided one does the calculations for both cases correctly using knowledge of pressure. Again, this is the beauty of science theories that are proven to be correct almost all the time unless refuted by newer findings and discoveries.
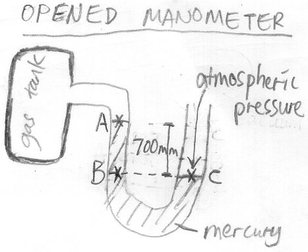
Qn 3: Find the gas pressure in the gas tank.
Note that pressures at same level are always the same (after a while, one should get used to this pressure concept).
Thus, PB = PC
PA + Pressure difference between A and B = PC
PA + hρg = 1.03 X 10^5 , Pressure difference between A and B is given by hρg while PC is atmospheric pressure
PA + (0.7)(13600)(10) = 1.03 X 10^5
PA = 1.03 X 10^5 - 95200
= 7800 Pa
= 7.8 kPa
We see that the gas pressure in this gas tank is 7.8 kPa which is less than atmospheric pressure of 1.03 X 10^5 Pa. That is why the mercury in right arm of this U-tube is lower level than left arm of U-tube as the atmospheric pressure exerts greater pressure on the mercury surface in right arm than this gas of lower pressure exerting its pressure on mercury surface in left arm.
Note that pressures at same level are always the same (after a while, one should get used to this pressure concept).
Thus, PB = PC
PA + Pressure difference between A and B = PC
PA + hρg = 1.03 X 10^5 , Pressure difference between A and B is given by hρg while PC is atmospheric pressure
PA + (0.7)(13600)(10) = 1.03 X 10^5
PA = 1.03 X 10^5 - 95200
= 7800 Pa
= 7.8 kPa
We see that the gas pressure in this gas tank is 7.8 kPa which is less than atmospheric pressure of 1.03 X 10^5 Pa. That is why the mercury in right arm of this U-tube is lower level than left arm of U-tube as the atmospheric pressure exerts greater pressure on the mercury surface in right arm than this gas of lower pressure exerting its pressure on mercury surface in left arm.
Boyle's Law
Pressure of a gas is inversely proportional to the volume it occupies provided other factors such as temperature are kept constant.
P1V1 = P2V2
See following video for explanation of Boyle's Law and also a simple calculation using Boyle's Law.
P1V1 = P2V2
See following video for explanation of Boyle's Law and also a simple calculation using Boyle's Law.